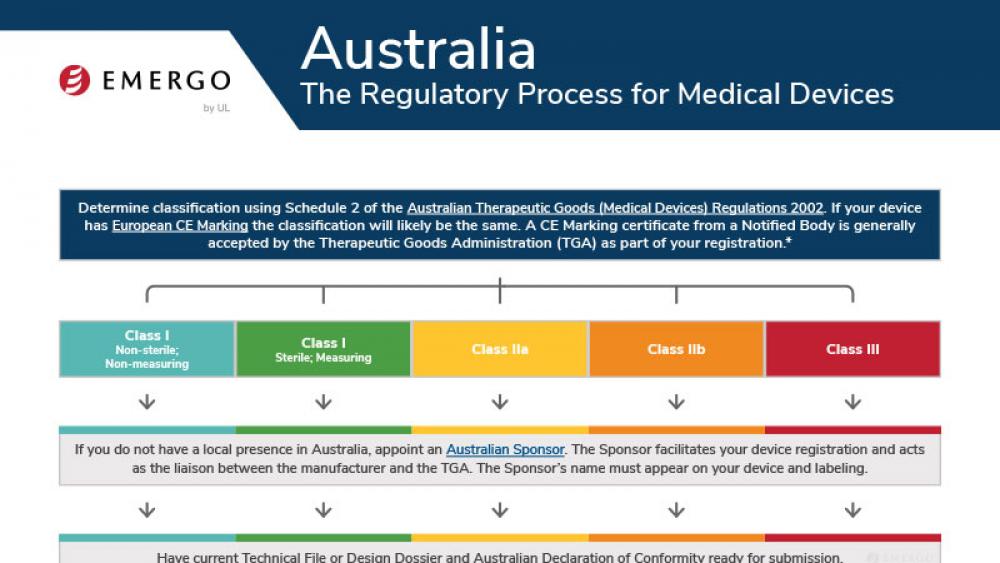
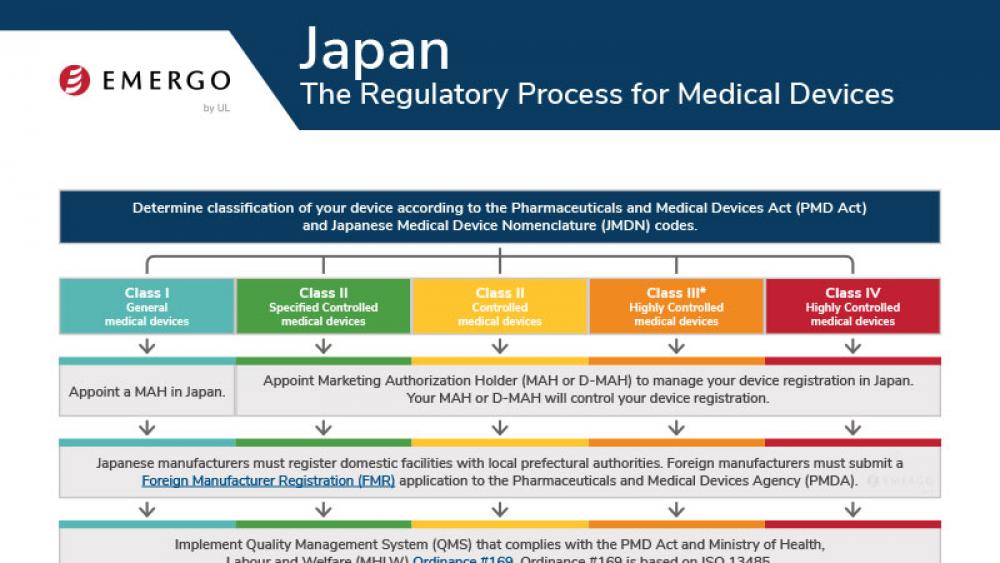
Australia | 医疗器械
澳大利亚TGA认证和医疗器械注册审批流程
This chart illustrates the steps in the Australia TGA medical device approval process and includes a timeline of expected approval.
2 pages
2013年 8月 29日
Brazil
巴西ANVISA医疗器械和IVD器械认证合规监管审批流程
This chart illustrates the steps in the Brazil ANVISA medical device approval process and includes a timeline of expected approval.
2 pages
2016年 7月 21日
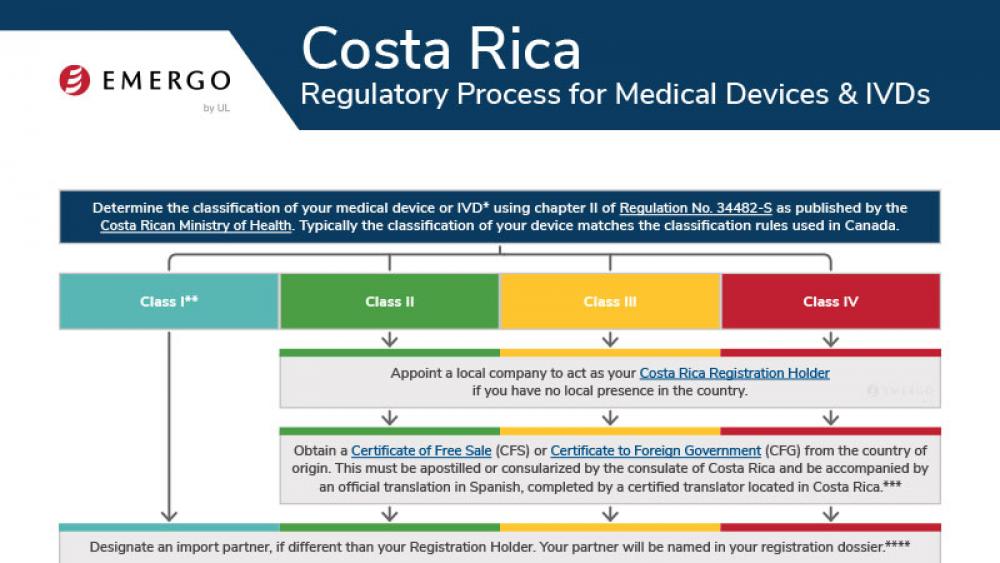
Costa Rica | 医疗器械
哥伦比亚医疗器械和IVD器械INVIMA认证合规审批流程
Medical device manufacturers who want to enter the Costa Rican market need to obtain approval from the Costa Rican Ministry of Health. There are a few pathways to approval, and documentation requirements are different depending on your device's class.
2 pages
2015年 6月 3日
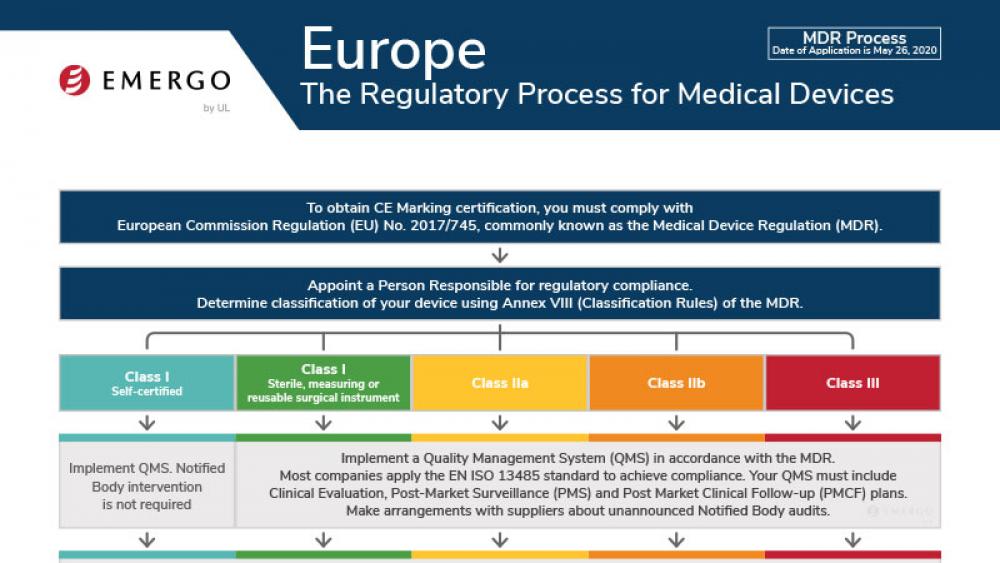
Europe | 医疗器械
欧盟医疗器械法规(MDR)CE认证标志合规流程
This chart illustrates steps to CE Marking in Europe's Medical Devices Regulation (MDR 2017/745) approval process.
2 pages
2017年 8月 23日
Saudi Arabia | 医疗器械
沙特医疗器械注册和SFDA认证合规审批流程
此流程图阐明了沙特阿拉伯每个医疗器械分类的注册和SFDA认证审批流程,可在法规事务管理平台(RAMS)下载。
2 pages
2013年 8月 29日
United States | 医疗器械
美国医疗器械FDA认证和体外诊断(IVD)器械注册流程
This chart illustrates the steps in the US FDA's medical device approval process and includes a timeline of expected approval.